You pick up your prescription refill, shake the bottle, and stare at the pills sitting at the bottom. They aren't right. Last month, they were round and yellow; today, they are oblong and white. Your first instinct might be panic: *Is this the wrong medicine? Did someone swap my script?* This reaction is completely understandable. For many people, the physical look of a pill is a vital part of the trust they place in their treatment.
If you've experienced this shift, take a breath. In the vast majority of cases, a change in how your medication looks does not mean a change in what it does. However, understanding why this happens helps ease the anxiety around your daily routine. The phenomenon is widespread. With generic drugs making up more than 70% of all prescriptions in the United States, visual variation is actually the norm, not the exception. Here is what you need to know about why your generic pills keep changing shape, whether it impacts your health, and how you can stay confident in your care.
The Business and Laws Behind Pill Shapes
To understand why your sertraline suddenly went from a green oval to a white diamond, you have to look at how the pharmaceutical industry operates under U.S. law. There is often a misconception that generic companies try to copy the famous brand name versions exactly. In reality, trademark laws actively prevent them from doing so. Trademark Law protects the unique identity of Brand Name Drugs, which includes the specific imprint, color, and shape chosen by the original manufacturer.
This legal framework ensures that two different products cannot look identical on store shelves. If a generic manufacturer produced a pill that looked exactly like the Pfizer version of Zoloft, they could be sued for infringement. As a result, each company producing a generic version must design its own distinctive packaging and pill characteristics. One pharmacy might supply bottles from Company A, while another month the supply chain shifts to Company B due to pricing contracts. Each company uses different molds, dyes, and tablet presses, leading to a rainbow of options for the exact same molecule.
Furthermore, pharmacies operate on thin margins. They automatically switch to whichever generic supplier offers the lowest cost at the moment. This economic decision-making happens behind the scenes, so the next time you walk in for a refill, the vendor might have changed. Dr. Darrick Lee from UCLA notes that once a branded medication becomes generic, various companies create their own versions. This isn't negligence; it's standard market behavior driven by the desire to keep costs low for insurance plans and patients.
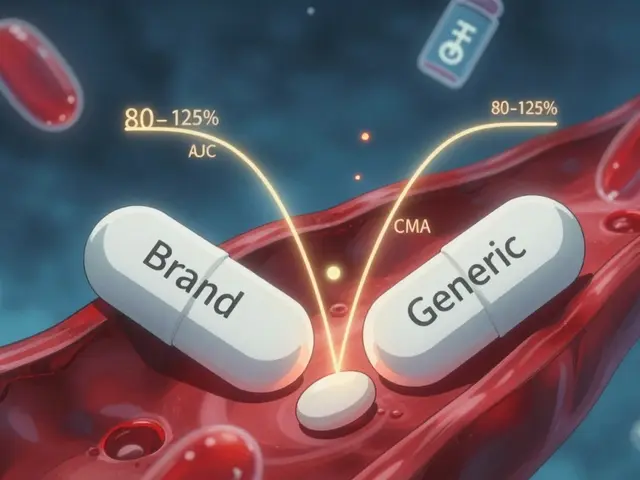
Is the Medicine Actually Different? The Science of Bioequivalence
The core question always remains: does a change in color mean a change in power? To answer this, we have to talk about how the regulatory bodies validate these drugs. The generic pill appearance standards set by federal agencies allow for cosmetic differences while strictly enforcing performance consistency.
The Food and Drug Administration (FDA) requires that any approved generic drug contain the exact same active ingredients as the brand-name counterpart. This active ingredient is the part of the pill that actually treats your condition-like the lithium in mood stabilizers or the lisinopril in blood pressure meds. Regardless of whether the pill is pink or peach, the amount of active chemical per tablet must match the original brand standard.
However, the "inactive ingredients" can vary. These are fillers, binders, and coloring agents used to hold the pill together or give it a specific hue. While usually harmless, these are the components responsible for visual changes. Some fillers might affect digestion slightly differently, such as lactose intolerance issues, but the therapeutic effect remains the same. This concept is known as bioequivalence.
| Feature | Brand Name Drug | Generic Drug |
|---|---|---|
| Active Ingredient | Must be identical | Must be identical |
| Dosage Strength | Fixed standard | Fixed standard |
| Color & Shape | Protected Trademark | Varies by manufacturer |
| Price | Higher (Patent protected) | 80-85% Lower |
When a generic drug is approved, it has to prove through rigorous testing that it releases the active ingredient into the bloodstream at the same rate and to the same extent as the brand version. This ensures that the new pill will work in your body just as effectively as the old one, regardless of its physical form. So, scientifically speaking, the difference lies in the wrapper, not the weapon.

The Human Cost of Visual Confusion
While science confirms the safety, the human experience tells a different story. Our brains rely heavily on pattern recognition. When we take medication every day, we form a visual anchor. That anchor provides reassurance. When the pill changes, it disrupts the routine and triggers uncertainty. Studies suggest this confusion can be dangerous not because the drug is bad, but because the patient stops taking it.
Data published in the Annals of Internal Medicine highlights a stark reality. Research indicates there is a 34% likelihood of a patient discontinuing medication after a color change alone. That number jumps to a staggering 66% likelihood after a shape change. Imagine a patient managing heart failure who relies on diuretics. If their trusted small red pill becomes a large white capsule, they might suspect it's an error and simply put it aside.
Dr. Erin M. Noren, an internal medicine physician, emphasizes the psychological aspect of this issue. Patients often associate certain colors with effectiveness. A distinct neon orange tablet might feel "stronger" than a plain white one. When that connection is broken, trust erodes. This non-adherence puts lives at risk more than the actual formulation ever would. Misidentification can also lead to accidental overdose if a patient thinks the new pill is a different, stronger drug meant for an emergency rather than their daily dose.

Actionable Steps for Patients and Caregivers
If you are the one opening the bottle, what can you actually do to bridge this gap between pharmacy logistics and your peace of mind? You shouldn't have to navigate these uncertainties alone. Pharmacists are trained resources, but sometimes you need to be proactive about your own record-keeping.
- Maintain a Personal Medication List: Keep a written or digital log of every medication you take. Include the name, dosage, and ideally, a description of the last appearance (color, shape, imprint code). Bring this list to every visit. It helps doctors verify what you are supposed to be taking versus what is physically in front of you.
- Ask for Identification Codes: Every legitimate pill has an imprint code-a letter or number stamped onto the surface. Write down the letters on your current bottle. If the pill changes next month, compare the new imprint code online to confirm it matches the drug name.
- Talk to Your Pharmacist Immediately: Do not swallow a suspicious-looking pill until you ask. Tell them, "This doesn't look like last month." If the change makes you uncomfortable, ask if they can specifically request a product from the manufacturer whose pills you prefer. While they may charge slightly more to find a specific batch, some pharmacies can lock your prescription to a preferred source.
- Utilize Online Tools: Resources like the Medscape Pill Identifier are incredibly useful. Snap a photo or describe the shape/color/imprint, and the database will tell you exactly what the pill is and what it contains.
- Check for Notification Stickers: Many modern pharmacies now place stickers on bottles indicating a change in manufacturer or appearance. About 78% of pharmacies included these notifications as recently as 2025. Check the label before popping the cap.
Regulation Updates and Future Trends
The healthcare system is aware of these friction points. Regulatory frameworks evolve to protect patients better without stifling the competition that keeps prices low. Recent legislative moves, such as the MODERN Labeling Act of 2020, have streamlined how labeling gets updated when new safety information emerges. More recently, proposed changes under Section 505(o)(4) of the Federal Food, Drug, and Cosmetic Act authorize stricter requirements for labeling updates based on new safety data.
There is also a growing conversation about standardization. While trademark laws exist, experts like Drs. Uhl and Peters have argued that having bioequivalent drugs look like their brand counterparts significantly enhances patient acceptance. Some states and pharmacy networks are beginning to advocate for fewer visual variations within a single geographic area to reduce confusion. However, until Congress alters the fundamental trademark protections, diversity in appearance will remain a feature of the U.S. market.
In 2025, analysis by the National Community Pharmacists Association noted that over 60% of independent pharmacies had implemented formal pill identification programs. This trend is likely accelerating. As AI integration grows in retail pharmacies, we might see automated scanning systems that notify you instantly when a dispensing batch differs visually from your previous one. Until then, awareness is your best defense. Knowing that the change is expected, legal, and safe allows you to move forward with confidence in your regimen.
Is it safe if my pill looks different?
Yes, generally it is safe. While the color and shape can change due to different manufacturers, the active ingredient and strength are required by the FDA to be identical to the brand name version.
Can I ask for the same pill color?
You can ask your pharmacist to dispense from a specific manufacturer that produces the look you prefer, though they may not always guarantee it for future refills based on inventory availability.
Why do generic pills change appearance?
Pill appearance changes occur because different generic manufacturers use different molds and dyes, and pharmacies switch suppliers to get the lowest price for your insurance plan.
Does a color change affect how the drug works?
No. The FDA requires generic drugs to demonstrate bioequivalence, meaning the new pill absorbs into your bloodstream at the same rate and level as the old one.
Should I throw away a pill that looks different?
Do not discard it yet. Verify the name, dosage, and imprint code with your pharmacist. If everything matches and only the appearance differs, you can safely continue taking it.

 How Effective Is Calcipotriene for Treating Nail Psoriasis?
How Effective Is Calcipotriene for Treating Nail Psoriasis?
 Bioequivalence Studies: What the FDA Requires Generic Drug Manufacturers to Prove
Bioequivalence Studies: What the FDA Requires Generic Drug Manufacturers to Prove
 The History of Bisoprolol Fumarate: From Discovery to Medical Use
The History of Bisoprolol Fumarate: From Discovery to Medical Use
 A Comprehensive Guide to Calcium Acetate Safety and Toxicity
A Comprehensive Guide to Calcium Acetate Safety and Toxicity
 Digital Therapeutics and Medication Interactions: What You Need to Know in 2026
Digital Therapeutics and Medication Interactions: What You Need to Know in 2026
Rachael Hammond
March 28, 2026 AT 22:23this is such a good explanation for what happens. u guys really need to read this cause most of thier minds go to fear when they see the change. i remember feeling anxious about my own sertraline pills last year when they came in a diffrent shape. the law part makes a lot of sense even if it feels weird sometimes. knowing the active ingredient stays the same helps me feel beter about the switch. it is important we trust the science behind these things. pharmacies work hard to keep costs low for everyone on insurance plans. we should give them the benefit of the doubt here. thanks for sharing all these facts about the manufacturing process. it clears up a lot of confusion for people like me who worry too much.
tyler lamarre
March 30, 2026 AT 08:43Surprised anyone thinks color determines efficacy when the molecule is the exact same thing. Most folks just dont want to learn how supply chains actually work. You should stop panicking over packaging details.
Devon Riley
March 30, 2026 AT 18:04It is so nice to see someone talk about this honestly.
I used to panic whenever my bottle looked different too.
My heart would race thinking i got the wrong meds.
Reading this helps calm my anxiety down though.
The science part explains everything perfectly well.
We forget that insurance drives the supply chain choices.
Pharmacies want the cheapest option for everyone always.
That makes sense why the color changes happen now.
My mom stopped taking hers because of the shape once.
She thought it was a mistake by the doctor office.
Luckily we called the pharmacist before she panicked.
They explained the bioequivalence thing to her calmly.
It saved me from worrying so much about refills.
We should all keep a photo log of our pills though.
Just so you know what is supposed to be inside.
Stay safe everyone out there reading this today. ❤️😊
Paul Vanderheiden
March 31, 2026 AT 02:29thanks for sharing your story it really helps me understand why my dad gets worried too. he always asks me about the pills and i tell him not to stress about the color. keep spreading awareness about this stuff because it saves lives. glad you found the info useful and we are all learning together here. 👍
Tony Yorke
April 1, 2026 AT 05:02Solid read.
kendra 0712
April 1, 2026 AT 21:01Wow this is so helpful!! Thank you for posting your experience!! Its scary when the meds change!!! I am definitely going to take pics now!! Keep being strong!! Love the advice!!
Tommy Nguyen
April 2, 2026 AT 10:34Good info here hope it helps people relax
Jeannette Kwiatkowski Kwiatkowski
April 2, 2026 AT 19:16Obviously the regulatory framework dictates these aesthetic variations. It is clear many readers misunderstand the trademark protections involved. One must consider the economic incentives driving the pharmacy vendors. Ignoring these factors leads to unnecessary patient anxiety. We need to trust the established quality control measures. The alternative creates chaos in healthcare delivery systems. Education is vital for proper medication adherence rates. Everyone should study the bioequivalence data closely. Personal preference rarely outweighs scientific validation protocols. It is imperative we maintain rational perspectives on generic substitutions.
Kameron Hacker
April 3, 2026 AT 03:30While these points on regulation are correct, patient psychology remains a critical factor we cannot ignore completely. Fear drives non-adherence regardless of statistical safety guarantees. We must acknowledge the emotional weight patients carry when routines are disrupted unexpectedly. A purely technical discussion fails to address the real world impact on health outcomes. Trust is fragile and visual consistency supports patient confidence significantly. Dismissing concerns as irrational ignores the complexity of human behavior patterns. We need balanced approaches that respect both law and mental health needs. Ignoring the human element leads to poor compliance statistics universally. Solutions should integrate education alongside regulatory clarity for best results. True progress requires listening to those who experience the confusion directly.
Poppy Jackson
April 3, 2026 AT 06:29Oh my goodness i nearly had a heart attack when i opened my box yesterday!!!! The white oval was nothing like the yellow round ones before!!!! It felt like a nightmare coming true right before my eyes!!!! I almost threw the bottle away without even asking anyone!!!! I am so glad i read this thread to save me from making a huge mistake!!!! Please everyone double check before you throw anything away!!!! It truly changed my mind about the whole situation today!!!!
Rohan Kumar
April 4, 2026 AT 18:49Big pharma knows exactly what they are doing with these changes 🤡 They think we wont notice they swapping ingredients slowly over time 🧐 The FDA approves it all so who cares about the little people 💀 Its funny how they expect us to just trust the labels blindly 🙄 I still suspect something fishy about the new batches honestly 🤨 We should all stay vigilant about these corporate tricks 🛑